My fertilisation protocol
And various nutrient digressions
For several years now, I have been applying a fertilisation protocol that is as simple as it is effective:
an exclusively organic-based fertiliser application in mid-autumn (right now, in the northern hemisphere), to be incorporated into the first layer of soil, or at least below the mulch (I get on well with pelleted manure) ;
a couple of administrations of synthetic fertiliser, in granule form, after the onset of the vegetative phase and in the middle of it (depending on how the season unfolds, around April and around June), to be spread directly on the soil.
Disclaimer: There is an extensive variety of plants, and this is a generalist approach that naturally doesn't account for specificities. Moreover, I define myself as a “pure” gardener, exclusively dedicated to ornamental plants. I seldom deal with edible plants, for which the subject of fertilisation is crucial, varied, and articulated at the same time. Let's just say that ornamental plants, in general, tend to be a bit more forgiving.
Now, I've covered the essentials, and I could end the article here... But how can I spare you the big explanation?
Let's start far, far away. Plants are autotrophic organisms capable of autonomously synthesizing glucose (C6 H12 O6), a fundamental molecule for cell metabolism, from water (H2O), carbon dioxide (CO2), and sunlight through chlorophyll photosynthesis, an elementary process known even to primary school children (I was nine or ten years old, a substitute teacher came to school and mortified the class for our ignorance about photosynthesis. I still remember the dismay).
However, are these few elements sufficient for higher plants to survive and thrive? It's a rhetorical question with the answer being of course “no”. Not even simple single-celled algae can make it with just hydrogen (H), oxygen (O), and carbon (C).
In fact, plants require a whole array of nutrients that they must absorb from their environment (usually in the form of ions) to develop properly. These are macronutrients and micronutrients, subdivided based on whether they are required in greater or lesser quantities:
macronutrients: nitrogen (N), potassium (K), calcium (Ca), phosphorus (P), magnesium (Mg), sulfur (S).
micronutrients: iron (Fe), chlorine (Cl), manganese (Mn), boron (B), zinc (Zn), copper (Cu), molybdenum (Mo).
Please note that all nutrients are crucial for plant metabolism, and even minimal deficiencies in any of them immediately affect plant health.
Depending on their functions, macronutrients and micronutrients can be further subdivided into structural nutrients (C, H, O, N, P, S, Ca, and Mg), which play a role in constructing plant tissue, and nutrients with metabolic and physiological functions. For example, the “heart” of chlorophyll, perhaps the most important pigment in the photosynthetic process, consists of a magnesium atom (Mg) bound to four nitrogen atoms (N): definitely structural nutrients!
![Picture credits: [Chlorin ring] (n.d.). [picture]. https://users.highland.edu/~jsullivan/principles-of-general-chemistry-v1.0/s25-05-the-s-block-elements-in-biolog.html. Picture credits: [Chlorin ring] (n.d.). [picture]. https://users.highland.edu/~jsullivan/principles-of-general-chemistry-v1.0/s25-05-the-s-block-elements-in-biolog.html.](https://substackcdn.com/image/fetch/$s_!4Qmd!,w_1456,c_limit,f_auto,q_auto:good,fl_progressive:steep/https%3A%2F%2Fsubstack-post-media.s3.amazonaws.com%2Fpublic%2Fimages%2F654d0f81-8dff-4ee3-afd5-b629904bbe48_857x469.jpeg)
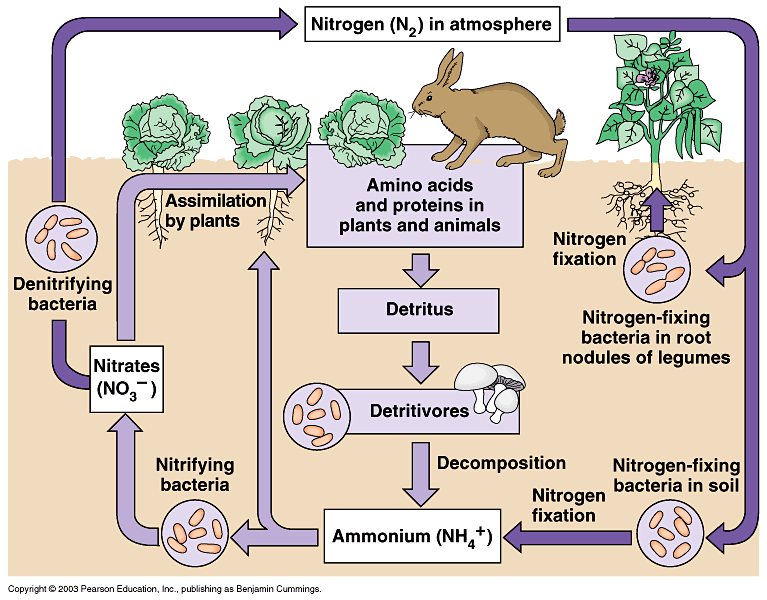
In ecology, we study how each chemical element of matter within an ecosystem follows a closed pathway, a cycle. Consider, for example, the element nitrogen (N). It is commonly found in gaseous form and makes up most of the air we breathe. Thanks to the action of specific bacteria called “nitrogen-fixers”, nitrogen is transformed into compounds assimilable by plants, which integrate it into their tissues. Some plants are then eaten by animals, so the nitrogen continues its path in the ecosystem. When the organisms die, the nitrogen compounds return to the soil, and after a series of steps, the nitrogen is once again found in the atmosphere, in gaseous form.
In an artificial cultivation context such as gardens or, even worse, agriculture, the natural cycles are incomplete, interrupted, or completely absent, and one can therefore incur a slow and progressive depletion of macro- and micronutrients that tends to impoverish the soil: in essence, the plants assimilate them, but the conditions for them to return to the soil hardly ever occur. This is why fertilisation is necessary: it is, in fact, crucial to reintroduce into the soil the nutrients that are gradually subtracted with the aim of maintaining soil fertility, rather than increasing it.
Fertilisation can be either natural and takes place by adding organic fertiliser consisting of manure and decomposed plant residues to the soil (compost), or artificial, by administering synthetic inorganic salts (granules) to the soil, which dissolve in water and release the nutrient ions needed by the plant.
Both forms of fertilisation have advantages and disadvantages, which explains why I use both: in natural fertilisation, the nutrients become available to the plants only after a slow process of mineralisation of the organic matter by the soil micro-organisms (this explains why I feed organic fertilizer in the autumn: the nutrients will be available by early spring), whereas in artificial fertilisation they are made available to the plants immediately.
On the other hand, synthetic fertilisers do not enrich the soil with humus (the dark organic matter in soil that is formed by the decomposition of plant and animal matter) and thus cannot contribute to improving its structure and quality. Moreover, the nutrients released by the organic salts they are composed of tend to be easily washed away by water, while residues of these same salts can accumulate in the soil in certain contexts, resulting in hyper-salinization that proves extremely harmful to plants.
That’s all. True, these are familiar topics for those who are passionate about gardening, but sometimes it is good to refresh one's memory... Especially to myself.